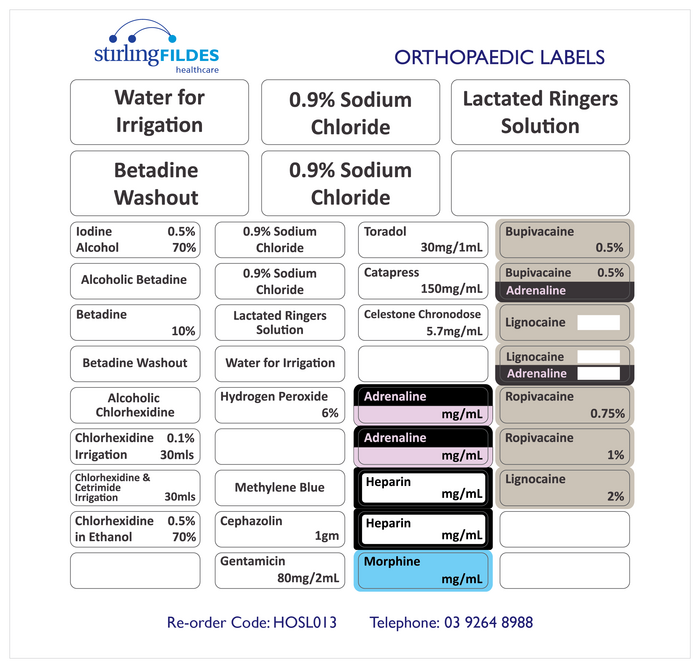
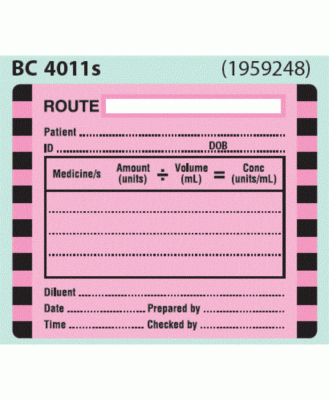
42 cautionary and advisory labels for medicines
› Details › F2019C00136Therapeutic Goods Regulations 1990 - Legislation Feb 12, 2019 · Required Advisory Statements for Medicine Labels means the advisory statements specified by the Minister by legislative instrument under subsection 3(5A) of the Act. sample includes part of a sample. serious, in relation to a form of a disease, condition, ailment or defect, means a form of the disease, condition, ailment or defect that is: COVID-19 regulatory changes | Pharmaceutical Society of ... 31/03/2022 · Pharmacists are also reminded that APF25 recommends cautionary advisory labels 21 and A for estradiol tablets due to the potential risk of harm through unintended exposure. Advice should be provided, if relevant, to patients, carers and healthcare workers on how to avoid unintended exposure to estradiol. Use professional judgement on whether label A …
Dispensing Practice Guidelines 2019 JUNE Cautionary advisory labels 22 Supplying and counselling 23 Final checking 23 Correctly identifying the patient 23 Patient counselling 23 Delegation and supervision in the dispensing process 25 Supervision of dispensary assistants/technicians 25 Risk management 26 Standardised dispensing process 26 Workloads 26 Incident management 27 Standard …

Cautionary and advisory labels for medicines
› en › health-canadaGuidance Document: Labelling of Pharmaceutical Drugs ... - Canada Nov 01, 2013 · The purpose of this document is to provide guidance to sponsors to facilitate compliance with the labelling requirements pursuant to sections 3, 9, and 10 of the Food and Drugs Act as well as related provisions of the Food and Drug Regulations, the Controlled Drugs and Substances Act, and its related Regulations including the Narcotic Control Regulations, Parts G and J of the Food and Drug ...
Cautionary and advisory labels for medicines. › en › health-canadaGuidance Document: Labelling of Pharmaceutical Drugs ... - Canada Nov 01, 2013 · The purpose of this document is to provide guidance to sponsors to facilitate compliance with the labelling requirements pursuant to sections 3, 9, and 10 of the Food and Drugs Act as well as related provisions of the Food and Drug Regulations, the Controlled Drugs and Substances Act, and its related Regulations including the Narcotic Control Regulations, Parts G and J of the Food and Drug ...

A selection of warning labels is available at a Southern California hospital pharmacy to ensure ...










Post a Comment for "42 cautionary and advisory labels for medicines"